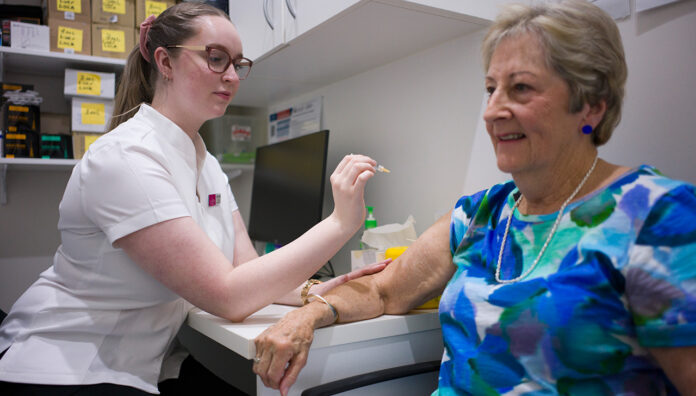Last week, the Australian Technical Advisory Group on Immunisation (ATAGI) announced vaccine recommendations for three respiratory conditions – influenza, COVID-19 and Respiratory syncytial virus (RSV).
With vaccine advice for multiple respiratory conditions released simultaneously, co-administration should be a big focus for pharmacists this season, said Chris Campbell MPS, PSA General Manager Policy and Program Delivery.
‘If someone attends the pharmacy for an influenza vaccine, now is the opportunity to see if they are eligible for a COVID-19 booster, whether the RSV vaccine appropriate for them if they are over 60; as well as a raft of other National Immunisation Program (NIP) immunisations,’ he said.
‘They may come in for one vaccine, but for many people, they could be eligible for three to five eligible vaccines, many of which will be NIP and NIPVIP funded.’
Here are the key changes pharmacists need to be aware of this season.
COVID-19
This year, ATAGI’s 2024 COVID-19 vaccination recommendations finally provide clarity for both pharmacists and the public on the 2024 booster program, said Mr Campbell.
Booster doses are now recommended twice a year for people over the age of 75 years. Other at-risk individuals are now recommended to have an annual booster every 6 or 12 months, and all people over 18 years can have a COVID–19 booster annually if they choose.
There have also been updates to the primary dose recommendations, including removal of the routine recommendation for children to receive a primary course of the vaccine. Now, only at risk children are recommended to receive a primary course of vaccine.
‘These updates are designed to reflect changes to not just the circulating virus, but also herd immunity, said Mr Campbell.
RSV
Following the release of ATAGI’s RSV vaccine recommendations, NSW became the first jurisdiction to change the legislative instrument, updating the NSW Pharmacist Vaccination Standards so pharmacists can initiate and administer RSV vaccines.
The NSW standards were also updated to include the pneumococcal conjugate vaccine for people aged 70 years and over, and Aboriginal and Torres Strait Islander people aged 50 years and over.
PSA NSW President Luke Kelly FPS said it was important for people to protect themselves against infectious diseases such as RSV ahead of winter.
‘We’ve seen massive advances in vaccine technology in recent years and it’s fantastic older Australians – the people who are most at risk of complications and death from RSV – can take positive steps to protect themselves,’ he said.
ATAGI has recommended RSV vaccination for:
- patients aged 75 years and older
- Aboriginal and Torres Strait Islander people aged over 60 years
- patients over 60 who have a medical condition that increases the risk of severe disease due to RSV.
Medical conditions associated with an increased risk of RSV disease complications for which RSV vaccination is recommended in adults ≥60 years
| Category | Example medical conditions |
|---|---|
| Cardiac disease | Congenital heart disease, congestive heart failure, coronary artery disease |
| Chronic respiratory condition | Suppurative lung disease, bronchiectasis, cystic fibrosis, chronic obstructive pulmonary disease, chronic emphysema, severe asthma (requiring frequent medical consultations or the use of multiple medicines) |
| Immunocompromising condition | HIV infection, malignancy, immunocompromise due to disease or treatment, asplenia or splenic dysfunction, solid organ transplant, haematopoietic stem cell transplant, CAR-T cell therapy |
| Chronic metabolic disorder | Type 1 or 2 diabetes, amino acid disorders, carbohydrate disorders, cholesterol biosynthesis disorders, fatty acid oxidation defects, mitochondrial disorders, organic acid disorders, urea cycle disorders, vitamin/cofactor disorders, porphyria |
| Chronic kidney disease Stage 4 or 5 | |
| Chronic neurological condition | Hereditary and degenerative central nervous system diseases, seizure disorders, spinal cord injuries, neuromuscular disorders, conditions that increase respiratory infection risk |
Note: These examples are not exhaustive, and providers may include individuals with conditions similar to those listed above based on clinical judgement.
However, all patients over 60 years of age are eligible for RSV vaccination.
Mr Campbell indicated that it’s likely other jurisdictions will soon follow NSW lead by allowing pharmacists to administer the RSV vaccine.
‘The state and territory governments will need to update their legislative instruments for RSV vaccination, and it makes sense that they do it for all vaccine providers in the one go,’ he said.
‘So we may see a more rapid adoption of pharmacists being able to administer the RSV vaccine than any other new vaccine.’
The RSV vaccine is not yet funded under the NIP, and is predicted to remain on the private market throughout the 2024 season, said Mr Campbell. But this could also soon change.
‘For example, with the most recent shingles vaccine included on the NIP, which we saw enter the private market first, there was an appetite from patients to be protected from shingles,’ he said. ‘We suspect we’ll see the same demand for adults who want to protect themselves against RSV.’
However, given much of the population’s awareness of RSV is associated with children, pharmacists will need to actively discuss the risk of infection, and the benefits of vaccination to ensure uptake.
‘Awareness of the condition is still growing,’ said Mr Campbell. ‘This uniquely places pharmacists in a position to have that conversation with someone about whether the vaccine is going to be right for them.’
Influenza
This influenza season is already shaping up to be a big one. In January, national influenza cases were the highest they have been in 6 years, peaking at 9,685 cases followed by a dip in February to 6,344 cases.
‘This year, there’s a new vaccine now covered under NIP for eligible people aged 5-64 years,’ said Mr Campbell. ‘So it’s important for pharmacists to know which vaccine will be right for their patient.’
Seasonal influenza vaccines registered and available for use in Australia in 2024, by age
| Registered age group/vaccine | 6 months to <5 years | ≥5 to <60 years | ≥60 to <65 years | ≥65 years |
|---|---|---|---|---|
| Vaxigrip Tetra 0.5 mL (Sanofi) | ✓ available for free under the NIP | ✓ * available for free under the NIP | ✓ * available for free under the NIP | ✓ |
| Fluarix Tetra 0.5 mL (GSK) | ✓ available for free under the NIP | ✓ * available for free under the NIP | ✓ * available for free under the NIP | ✓ |
| Flucelvax Quad 0.5 mL (Seqirus) | ✓ | ✓ * available for free under the NIP | ✓ * available for free under the NIP | ✓ |
| Afluria Quad 0.5 mL (Seqirus) | X | ✓ | ✓ | ✓ |
| FluQuadri 0.5 mL (Sanofi) | ✓ | ✓ | ✓ | ✓ |
| Influvac Tetra 0.5 mL (Viatris) | ✓ | ✓ | ✓ | ✓ |
| Fluad Quad 0.5 mL (Seqirus) | X | X | X | ✓ available for free under the NIP |
| Fluzone High-Dose Quad 0.7 mL (Sanofi) | X | X | ✓ | ✓ |
* NIP funding only for Aboriginal and Torres Strait Islander people, pregnant women and people who have certain medical conditions.
Flucelvax Quad, the first cell-based influenza vaccine, is available on the NIP. While there is no preferential recommendation between the cell-based vaccine Flucelvax Quad and standard dose of egg-based influenza vaccines, Mr Campbell said the inclusion gives people an alternative vaccine choice.
‘An egg allergy hasn’t necessarily been the main reason for someone to not get the vaccine, because they cleave off the protein so well,’ he said.
‘However, there’s still scenarios where it is not appropriate and this provides another option.’
PSA supporting pharmacist vaccinators
To support members, PSA is holding briefings for each state and territory to explain these ATAGI changes, as well as state-specific application of the NIP, NIPVIP program rules and the updated vaccine recommendations.
- ATAGI Update (COVID-19, influenza, RSV), Tuesday 5 March 8:00 pm
‘We’ve invited representatives from the health departments, so pharmacists can ask them questions around how the programs work in each state and territory,’ said Mr Campbell.
‘To be ready to support your patients this winter, pharmacist immunisers should go to both the ATAGI and their state-specific member briefings.’
Dates releases for the state-based briefings include (local time):
- Queensland, Monday 4 March 7:00 pm
- Tasmania, Tuesday 5 March 7:00 pm (immediately prior to the member briefing on the ATAGI updates)
- NSW, Thursday 7 March, 7:00 pm
- Western Australia, Tuesday 12 March 7:00 pm
- Victoria, Tuesday 19 March 7:00 pm.
PSA is also hosting a webinar on Wednesday 20 March 7:00 AEDT on Respiratory Syncytial Virus Prevention in Older Adults Amidst Expanding Vaccine Landscape. Register here to attend.



 Jess Hadley, community pharmacist and Professional Officer at PDL[/caption]
Jess Hadley, community pharmacist and Professional Officer at PDL[/caption]
 Peter Guthrey, Senior Pharmacist – Strategic Policy at PSA[/caption]
Peter Guthrey, Senior Pharmacist – Strategic Policy at PSA[/caption]

 Professor Margie Danchin[/caption]
Professor Margie Danchin[/caption]
 Dr Peter Tenni[/caption]
Dr Peter Tenni[/caption]
 How should we deprescribe gabapentinoids, according to the Maudsley Deprescribing Guidelines[/caption]
How should we deprescribe gabapentinoids, according to the Maudsley Deprescribing Guidelines[/caption]