ADVERTORIAL BY BOEHRINGER INGELHEIM
Australia’s only soft mist inhaler, Respimat, is now available as a reusable device and is included on the PBS for asthma or COPD patients using Spiriva Respimat (tiotropium) or Spiolto Respimat (tiotropium/olodaterol).1-3
The new-generation Respimat inhaler has enhanced features and can be used with up to six cartridges, rather than being disposed of and replaced each time the medication runs out.4-5
Each year, 1.7 million fewer inhalers could be prevented from entering Australian landfill as existing Spiriva and Spiolto Respimat patients transition to the reusable inhaler.6
Discuss the transition to, and use of, the Respimat reusable inhaler with patients.
Instructional videos are available at: www.respimat.com.au (HCP password: softmist)
References
- Department of Health. PBS Schedule. 1 March 2021.
- Spiriva Respimat Approved Production Information. June 2020
- Spiolto Respimat Approved Production Information. November 2020
- Spiriva Respimat Instruction For Use.
- Spiolto Respimat Instruction For Use.
- Data on File. Boehringer Ingelheim.


Please review Product Information before prescribing. Full Product Information is available at www.boehringer-ingelheim.com.au/PI . Further Information is available from Boehringer Ingelheim Pty Ltd.
INDICATIONS: SPIRIVA RESPIMAT: Long term maintenance treatment of bronchospasm and dyspnoea associated with chronic obstructive pulmonary disease (COPD). Prevention of COPD exacerbations. SPIOLTO RESPIMAT: Once-daily maintenance bronchodilator treatment to relieve symptoms in adult patients with COPD. CONTRAINDICATIONS: SPIRIVA RESPIMAT and SPIOLTO RESPIMAT: Hypersensitivity to tiotropium bromide, olodaterol (Spiolto only), atropine or its derivatives, or to any of the excipients. PRECAUTIONS: SPIRIVA RESPIMAT and SPIOLTO RESPIMAT: Should not be used: more frequently than once daily; for relief of acute symptoms, treatment of acute episodes of bronchospasm, immediate hypersensitivity reactions, paradoxical bronchospasm, narrow-angle glaucoma, prostatic hyperplasia, bladder neck obstruction, urinary retention, micturition difficulties, recent myocardial infarction (<6 months for SPIRIVA, <12 months for SPIOLTO), unstable or life-threatening cardiac arrhythmia within past year, hospitalisation for heart failure within past year, moderate to severe renal impairment(CrCL ≤50 mL/min), pregnancy, lactation and children. Avoid solution or mist entering eyes. SPIRIVA RESPIMAT: Should not be used for: first-line treatment for asthma, dry mouth. SPIOLTO RESPIMAT: Should not be used: in treatment of asthma (LABAs may increase the risk of asthma-related hospitalisations and death); initiated in acutely deteriorating COPD, severe hepatic impairment, convulsive disorders, thyrotoxicosis, QT interval prolongation, unusual responsiveness to sympathomimetic amines; increases in pulse rate, blood pressure and/or symptoms of clinically significant cardiovascular effect, paroxysmaltachycardia (>100 beats per minute), hypokalaemia, hyperglycaemia. INTERACTIONS: SPIRIVA RESPIMAT and SPIOLTO RESPIMAT: Co-administration with anticholinergic drugs. SPIOLTO RESPIMAT: Co-administration with adrenergic agents, xanthine derivatives, steroids, non-potassium paring diuretics, beta-blockers, MAO inhibitors, tricyclic antidepressants, QTc interval prolonging drugs, LAMAs, LABAs. ADVERSE EFFECTS: SPIRIVA RESPIMAT: Common: Dry mouth, usually mild. SPIOLTO RESPIMAT: Very common: nasopharyngitis. Common: pneumonia, bronchitis, influenza, urinary tract infection, sinusitis, cough, dyspnoea, back pain, overdose, headache. Others, see full PI. DOSAGE: SPIRIVA RESPIMAT: For oral inhalation. 5μg tiotropium given as two puffs once daily, at the same time each day. SPIOLTO RESPIMAT: For oral inhalation. 5μg tiotropium and 5 μg olodaterol given as two puffs once daily, at the same time each day. Do not exceed recommended dose. Cartridges to be used only with RESPIMAT inhaler. March 2020
RESPIMAT®, SPIRIVA® and SPIOLTO® are registered trademarks of Boehringer Ingelheim. Boehringer Ingelheim Pty Ltd. ABN 52 000 452 308. 78 Waterloo Road, North Ryde, NSW 2113. PC-AU-101906. February 2021.


 Professor Margie Danchin[/caption]
Professor Margie Danchin[/caption]
 Dr Peter Tenni[/caption]
Dr Peter Tenni[/caption]
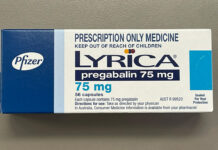
 How should we deprescribe gabapentinoids, according to the Maudsley Deprescribing Guidelines[/caption]
How should we deprescribe gabapentinoids, according to the Maudsley Deprescribing Guidelines[/caption]



 Pharmacists have always prescribed, but they have the potential to prescribe much more
Pharmacists have always prescribed, but they have the potential to prescribe much more







